"I say yes to participating in research because it gives access to a treatment that might otherwise not be available. And it feels good to know that, even in a small way, I can help.”
– Emily*, Richmond
The first day after a long bout of depression, Emily* did something she hadn’t done since she was a child—she grabbed her skimboard and headed down to Spanish Banks beach to take advantage of the low tide.
“I had to pick my husband up off the ground,” joked the 61-year-old. “He couldn’t believe it. I couldn’t believe it myself really. And, I can tell you that trying my hand at skimboarding again was not like riding a bicycle. But I had so much fun!”
The joy Emily and her husband felt that day was held against the backdrop of years living with a disease that can put moments like these out of reach.
Around 30 per cent of patients like Emily, who live with treatment resistant depression, do not know the root cause of their symptoms. Medications and psychotherapy can help, but often lose their effectiveness or stop working entirely after a while. And there is currently no cure for the illness.
For Emily, three decades of managing her depression has meant not always being able to work full-time as a nurse, and spending days unable to do the things she enjoys, such as gardening, going for coffee and walks with friends and looking up at the stars with her husband.
“When I am depressed, I feel no sense of enjoyment, no motivation, no appetite, nothing matters and I cannot feel affection towards others.”
Then, while in the midst of a recent depressive episode, a friend emailed Emily a recruitment poster for a clinical trial that was investigating an alternative procedure for treatment resistant depression.
Synching together parts of the mind
Emily participated in the clinical trial led by Dr. Fidel Vila-Rodriguez, a Vancouver Coastal Health Research Institute clinician-scientist and the director of the Non-Invasive Neurostimulation Therapies (NINET) Laboratory. The clinical trial examined an emerging treatment for mental health disorders called repetitive transcranial magnetic stimulation (rTMS) therapy.
The Health Canada- and FDA-approved treatment involves applying coils to the outside of the head that transmit magnetic field pulses to stimulate parts of the brain involved in emotional regulation.
Study participants each underwent five 30-minute treatments per week for four weeks, and followed up with clinicians after one week, four weeks and 12 weeks. They also received functional magnetic resonance imagining (fMRI) scans, which track blood flow in the brain.
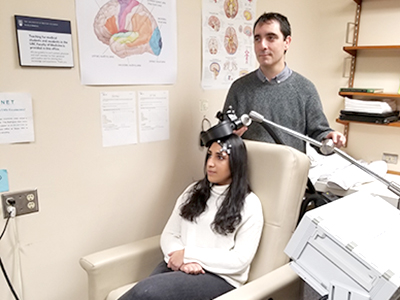
“For our brain to function properly, its different parts need to work together, almost like instruments in an orchestra,” says Vila-Rodriguez.
“Depression seems to throw off the cadence of our brains, but rTMS acts like a conductor who reestablishes its harmony.”
rTMS has been shown to reduce symptoms of depression by half for around 50 per cent of patients; and, around 30 per cent of patients have reported an absence of any symptoms1. The effect of rTMS can last for 12 months in many cases2. rTMS also has relatively few side effects—the chief complaints often being headaches and pain at the place of stimulation. However, it is not associated with weight gain or sexual dysfunction—as can be the case with medications—or with memory loss, which has been associated with electroconvulsive therapy.
Since completing her rTMS sessions through the clinical trial, Emily has started to feel better. She says her anxiety has decreased, her appetite has improved and she was able to appreciate spending time with her cat again.
“Around the end of my fourth week of treatment, I was feeling well enough to suggest to my husband that we start planning our trip to France, which we had postponed several months earlier because of my depression.”
“My husband tells me that my mood seems to be better and that I’m laughing more now.”
If she keeps feeling this good, Emily says she might be able to reduce her medication, and hopefully for a long time.
“Right now I am enjoying taking each day as it comes,” she says. “I’m very grateful to the research team, my care providers and my friends and family who have been so supportive. I know it’s not easy supporting someone who’s suffering from depression, but they have always made my difficult times much more tolerable.”
Participants are being recruited for this research study. For more information, contact research coordinator Afifa Humaira at 604-822-7308 or ninet.lab@ubc.ca.
1 Effectiveness of Theta Burst Versus High-Frequency Repetitive Transcranial Magnetic Stimulation in Patients With Depression (THREE-D): A Randomised Non-Inferiority Trial
2 Durability of Antidepressant Response to Repetitive Transcranial Magnetic Stimulation: Systematic Review and Meta-Analysis
THIS IS ONE PATIENT’S STORY OF PARTICIPATING IN A CLINICAL TRIAL. YOUR EXPERIENCE MAY DIFFER. LEARN MORE ABOUT CLINICAL TRIALS BEFORE PARTICIPATING.
*Name has been changed to protect participant anonymity.