New images provide clues about COVID-19 virus mutations and their impact on vaccine efficacy.
Research from Vancouver Coastal Health Research Institute scientist Dr. Sriram Subramaniam produced molecular images of how mutations in the novel coronavirus, SARS-CoV-2, are driving infection. A global first-of-its-kind, the three-dimensional images of the N501Y spike protein mutation provide clues for future treatment interventions.
“We moved very quickly to identify the potential consequences of these variants as they emerged to get a better sense of what new variants may be coming down the pike and advise on whether present vaccines will be able to protect against them,” says Subramaniam.

His research, published in PLOS Biology in April 2021, examined the key mutation present in the B.1.1.7 variant, now known as Alpha. Believed to have originated in the United Kingdom, Alpha has been identified as being more virulent than the virus circulating the globe when the COVID-19 pandemic was first declared in March 2020.
Coronaviruses, as well as HIV and influenza viruses, use a spike protein on their surface to bind to and invade host cells through their angiotensin-converting enzyme 2 (ACE2) receptors, explains Subramaniam. COVID-19 hijacks host cells’ DNA to replicate itself and multiply, driving and spreading the infection.
The Alpha variant features an N501Y mutation on its spike protein, which the research team produced using genetic engineering to conduct their examination.
“Once the genome sequence of COVID-19 was discovered in early 2020, it became clear that understanding the structure of the spike protein was the key to destroying the mechanism it uses to infect hosts.”
Subramaniam and his research team visualized the structure of spike protein carrying the key N501Y Alpha variant mutation, which is over 100,000 times smaller than a pinhead, with cryo-electron microscopes. The state-of-the-art technology emits beams of electrons into cells and tissues at ultra-cooled “cryo” temperatures of around minus 196 degrees Celsius.
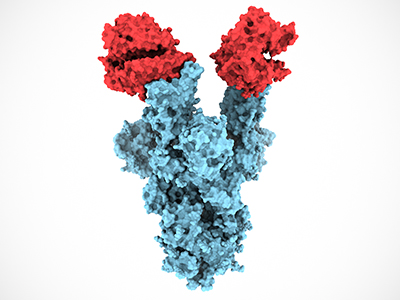
The 3D images of the N501Y mutation Subramaniam’s team obtained showed that the spike protein mutation is better at binding to host cells’ ACE2 receptors, which is likely what makes Alpha more virulent.
However, it was discovered that the overall structure of the spike protein was relatively unchanged, which should continue to enable current vaccine efficacy against Alpha, Subramaniam says.
Past virus research informs new insights into COVID-19
Before stepping into his current role at UBC, Subramaniam spent nearly a decade investigating HIV, influenza and Ebola viruses. This work provided him with insight into the unique challenges and advantages researchers and clinicians face in the search for the best treatments and preventative measures against COVID-19.
“One of the striking things about HIV is that the virus mutates at a very high rate,” notes Subramaniam. “Even within a single HIV patient, there could be dozens of strains of the virus circulating at any given time. This has made finding a vaccine for it challenging, even 40 years after it was first discovered.”
“In contrast, coronaviruses have historically not mutated at the same rate.”
“This gives COVID-19 vaccines a much higher chance of working; and, if enough people get vaccinated quickly, we can likely stay ahead of the mutation curve.”
A tricky aspect of the novel coronavirus is how it engages with the host, says Subramaniam. Many questions remain about why some people seem more prone to severe symptoms than others.
The images of the N501Y spike protein Subramaniam and his team collected are part of the global mission to fill in COVID-19 knowledge gaps, he says, and could “help researchers think about the next stage of COVID-19 vaccine design.”


